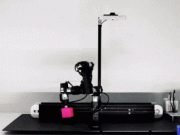
Researchers are always looking for ways to improve battery efficiency in robots. The University of Michigan engineers have now come up with a rechargeable zinc battery that integrates into the structure of robots to provide more energy. Here is how it works:
More like this ➡️ here
The new battery works by passing hydroxide ions between a zinc electrode and the air side through an electrolyte membrane. That membrane is partly a network of aramid nanofibers—the carbon-based fibers found in Kevlar vests—and a new water-based polymer gel. The gel helps shuttle the hydroxide ions between the electrodes.
Structural, rechargeable zinc battery
The battery is made of non-toxic materials. To test it, researchers experimented with worm and scorpion shaped robots.
[HT]
*Our articles may contain aff links. As an Amazon Associate we earn from qualifying purchases. Please read our disclaimer on how we fund this site.